| Description |
Taxanes are a class of molecules that are able to disrupt microtubule function, and thereby inhibiting cell proliferation. Hence, taxanes are commonly used in cancer treatment, often in combination with other anti-tumor drugs. However, low water solubility of taxanes present difficulties for drug development, while opting for organic solvents would cause severe toxicities in patients. Water-soluble prodrugs of the most common taxanes were developed for cancer treatment. These prodrugs show significantly higher bioavailability, and thus better in vivo drug activity. |
| Pipeline | Water-soluble prodrugs of the most common taxanes – LK-166 (cabazitaxel) and LK-196 (docetaxel) |
| Target | Tubulin |
| Drug Modality | Small molecule |
| Indication | Cancer |
| Product Category | Taxanes |
| Mechanism of Action | Inhibition of tubulin depolymerization to induce cancer cell apoptosis |
| Status | Preclinical |
| Patent | Granted |
All three water-soluble taxane prodrugs (LK-166 and LK-196) are currently open for out-licensing opportunities worldwide.
LK-166 (cabazitaxel) and LK-196 (docetaxel) are two water-soluble taxane prodrugs for cancer treatment, which have stable chemical properties and good water solubility.
Advantages:
● High drug safety – both prodrugs were developed without organic solvents.
● Higher efficacy – therapeutic window is significantly larger compared to the market competitors, thus producing greater therapeutic effect with increased dosing.
● Less blood toxicity at lower price – compared with albumin-bound paclitaxel.
● LK-166 can also be a potential treatment for refractory brain cancer.
Tubulin is a highly conserved αβ dimeric protein essential for all eukaryotes. α- and β-tubulins polymerize into microtubules, a major component of the eukaryotic cytoskeleton. Microtubules play a key role in many processes, including structural support, intracellular transport, and DNA segregation.
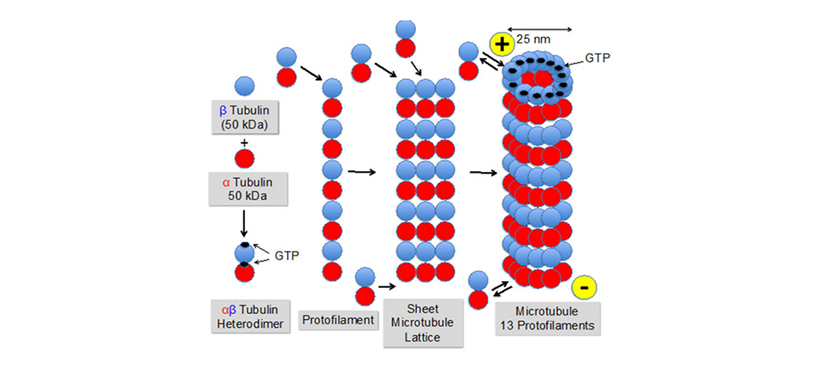
Figure 1. Formation of microtubules from α- and β-tubulin via αβ-heterodimers and protofilaments.
Krause. Cancer Drug Resist 2019; 2:82-106.
Specific isotypes of tubulin have been associated with cancer. The mutations of tubulin isotypes expressed in invasive tumors affect the binding of anti-cancer drugs and may contribute to drug resistance. For example, an increase in microtubule dynamicity has been found in cells which overexpress βIII-tubulin and may be related to the resistance of cancer cells to anti-microtubular drugs.
Cancer is a large group of diseases that can start in almost any organ or tissue of the body when abnormal cells divide uncontrollably, and invade other organs and tissues of the body to form metastatic cancer tumors.
Cancer is the second leading cause of death globally, accounting for an estimated 9.6 million deaths, or one in six deaths, in 2018. Lung, prostate, colorectal, stomach and liver cancer are the most common types of cancer in men, while breast, colorectal, lung, cervical and thyroid cancer are the most common among women. The cancer burden continues to grow globally, exerting tremendous physical, emotional and financial strain on individuals, families, communities and healthcare systems.
Tubulin-related inhibition is one of the targeting strategies in cancer treatment. Anti-tubulin agents that inhibit microtubule dynamics can be classified into: a) anti-depolymerization, and b) anti-polymerization of the microtubule cytoskeleton. Among microtubule depolymerization inhibitors, taxanes are considered as the most effective compounds, as they stabilize GDP-bound tubulin in the microtubule. As a consequence, the cell cycle of rapidly proliferating cancer cells comes to an arrest, eventually leading to apoptosis.
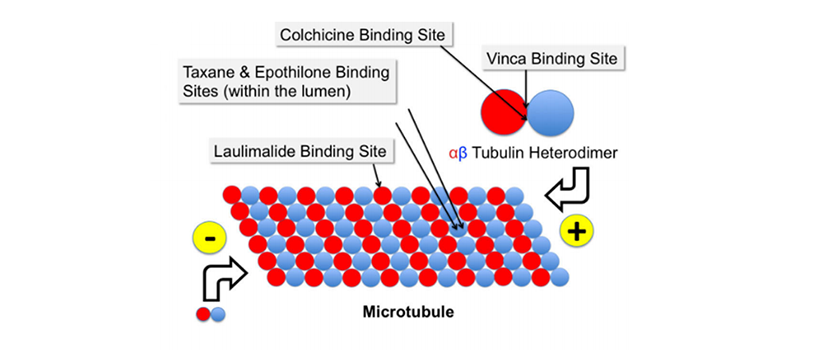
Figure 2. Binding sites for tubulin inhibitors.
Krause. Cancer Drug Resist 2019; 2:82-106.
LK-166 and LK-196 are currently in the preclinical stage. Patents for these products have been granted in the US and Europe; international patents under PCT are pending.
