| Drug Name | P75NEURO |
| Description |
Chronic neurodegenerative diseases such as Alzheimer’s and Parkinson’s diseases, as well as the acute conditions such as stroke and neurotrauma, degenerate the functions of the neuronal systems continuously as the diseases progress. The resulting social and economic burdens are enormous. To tackle the unmet needs, P75NEURO, a fusion protein drug that consists of the extracellular domain of p75 (p75ECD) and an immunoglobulin fragment C, was developed. P75NEURO prevents p75 neurothrophin receptor (p75NTR) from binding to various neurotrophic factors or nerve growth factors to antagonize the neurotoxic effects of p75NTR. Both in vitro and in vivo tests showed that P75NEURO was safe and efficacious with good druggability profile including good DMPK profile, stability in plasma (9 days of half-life), and no adverse effects at the effective dose. |
| Target | p75NTR (also named nerve growth factor receptor, NGFR) |
| Drug Modality | Fusion protein |
| Indication | Ischemic stroke; Alzheimer’s disease; Traumatic brain injury; Inflammatory pain |
| Product Category | Neurotrophic agent; Signal transduction modulator |
| Mechanism of Action | P75NEURO antagonizes the neurotoxic effects of the p75NTR. |
| Status | Preclinical |
| Patent | Patents on using P75NEURO for the treatment and diagnosis of neurological diseases have been filed. |
Protheragen Inc. is actively seeking partnership and licensing deals to further develop P75NEURO.
We look forward to collaborating with you soon.
| Introduction | The extracellular domain of the nerve growth factor receptor (NGFR) includes four 40-amino acid repeats with 6 cysteine residues at conserved positions followed by a serine/threonine-rich region, a single transmembrane domain, and a 155-amino acid cytoplasmic domain. The cysteine-rich region contains the nerve growth factor binding domain. |
| Approved Name | Nerve growth factor receptor |
| Official Symbol | NGFR |
| Gene Type | Protein coding |
| Synonyms | CD271; p75NTR; TNFRSF16; p75(NTR); Gp80-LNGFR |
| Ensembl | ENSG00000064300 |
| Gene ID | 4804 |
| mRNA Refseq | NM_002507 |
| Protein Refseq | NP_002498.1 |
| OMIM | 162010 |
| UniProt ID | P08138 |
| Chromosome Location | 17q21.33 |
| Gene Function |
p75NTR has 3 transcripts (splice variants), 245 orthologues, and 21 paralogues. |
| Pathway | p75NTR is a low affinity receptor that can bind to neuron growth factor (NGF), brain derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3), and neurotrophin-4 (NT-4). It mediates the neuronal cell survival and death. |
| Major Conditions | Neurological disorders; Cancer |
Neurological diseases, also known as neurological disorders or nervous system diseases, refer to a small class medical conditions affecting the nervous system. Stroke, Alzheimer’s disease, and traumatic brain injury all belong to this category. As a neuroprotective agent, P75NEURO can hinder or prevent the development of nervous system diseases by protecting the neurons from injuries. In addition, P75NEURO has potential therapeutic values for the cardiovascular disease and inflammatory pain.
| Ischemic stroke |
Stroke is a leading cause of morbidity and mortality on a global scale, a trend that is expected to continue as a result of the economic transition in the low- and middle-income countries. Referring to central nervous system infarction accompanied by overt symptoms, ischemic stroke accounts for almost 85% of all stroke events. According to the American Heart Association/American Stroke Association, total annual stroke-related costs are expected to reach USD 240.67 billion by 2030, a 129% increase over the cost at 2012. Acute intervention for ischemic stroke involves the administration of thrombolytic agents to restore perfusion and/or neuroprotective agents to limit neuronal damage. |
| Alzheimer’s disease |
Alzheimer’s disease (AD) is a slowly progressive and ultimately fatal degenerative brain disorder that primarily affects the elderly. AD is the most common cause of dementia and accounts for 60% to 80% of the cases. According to Lancet Commission’s estimate, approximately 47 million people worldwide were living with dementia, including AD, in 2017 and predicted that this number would triple by 2050. The financial costs attributable to AD and other forms of dementia are USD 818 billion globally in 2015. At 2018, the global social costs of dementia toped USD 1 trillion. While available therapies are often effective in palliating symptoms and improving quality of life, existing drugs do not target the underlying disease pathology and are not capable of prolonging survival. |
| Traumatic brain injury |
Traumatic brain injury (TBI) results from a sudden and violent impact to the head or when an object penetrates the skull and enters the brain tissue. It frequently occurs as a consequence of motor vehicle accidents, falls, sports injuries, or violent assaults. However, TBI is a complex and continuous disease process characterized by both structural damage and functional consequences. An international team has calculated a higher estimate of 69 million (range 64 to 74 million) episodes of TBI worldwide each year. TBI costs the global economy approximately USD 400 billion annually, equivalent to approximately 0.5% of the entire annual gross world product. A person suffering traumatic brain injury should receive immediate medical attention. Stabilization and treatment may prevent secondary injury. |
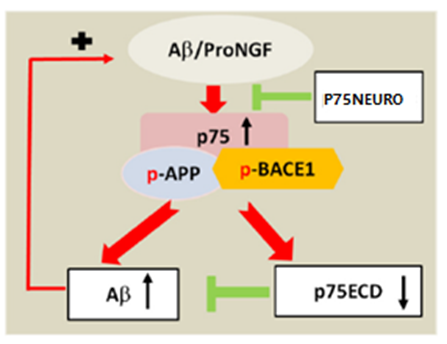
P75 neurotrophin receptor (p75NTR), a receptor for brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), and nerve growth factor precursor protein (proNGF), regulates the neuronal growth and death. P75NTR is also a receptor for β-amyloid (Aβ). The extracellular domain of p75 (P75ECD) is an endogenous neuroprotective protein that actively blocks the progression of neurodegeneration in neurological diseases. A dimer fusion protein consisted of P75ECD and Fc fragment of human immunoglobulin, P75NEURO inhibits the secretion of inflammatory cytokines and prevents the binding of p75NTR to Aβ and proNGF by:
● Blocking amyloidogenesis by preventing β-amyloid precursor enzyme-1 (BACE1) expression and endocytosis;
● Blocking tau hyperphosphorylation induced by both Aβ and proneurotrophins.
Patents on using P75NEURO for the treatment and diagnosis of neurological diseases have been filed in Australia and through patent cooperation treaty (PCT).
