| Drug Name | P11 |
| Description |
To achieve an ultra-long-acting and effective GLP-1 analogue of minimized side effects, a unique GLP-1 protection technology was developed to prevent degradation mediated by proteolytic enzymes. The modified GLP-1 analogues are gradually released in vivo, prolonging the effective duration of treatment. The commercially available Trulicity (developed by Eli Lilly) for treating diabetes is also a GLP-1 analogue, also resulting in hypoglycemic weight loss. Our In vivo and in vitro experiments have shown that applying our protection technology to Trulicity leads to a more constant drug concentration in blood, significantly longer half-life, and less side effects. In addition, the plan to develop a long-acting GLP-1/GIP dual agonist using this protection technology has been proposed. |
| Target | GLP-1 receptor |
| Drug Modality | Fc fusion protein; Polypeptide |
| Indication | Type 2 diabetes |
| Product Category | GLP-1 analogue |
| Mechanism of Action | As a GLP-1 analogue, P11 binds to GLP-1 receptors to activate intracellular signaling pathways, achieving a unique hypoglycemic effect that can be prolonged by this novel protection technology. |
| Status | Preclinical |
| Patent | Relevant patents submitted |
Protheragen Inc. is actively seeking partnership to further develop P11. Potential collaboration can be strategic alliance, licensing, or marketing agreement.
We look forward to hearing from you.
| Introduction |
Glucagon-like peptide-1 (GLP1) is a hormone derived from the preproglucagon molecule (GCG; 138030) and is secreted by intestinal L cells. It is the most potent stimulator of glucose-induced insulin secretion and also suppresses in vivo acid secretion by gastric glands. GLP1 receptor (GLP1R) bind to GLP1 with high affinity and the binding between the two activates adenylate cyclase. The receptor is 463 amino acids long and contains 7 transmembrane domains. Sequence homology is found only with the receptors for secretin, calcitonin, and parathyroid hormone, which together form a newly characterized family of G protein coupled receptors. |
| Approved Name | Glucagon like peptide 1 receptor |
| Official Symbol | GLP1R |
| Gene Type | Protein coding |
| Synonyms | GLP-1; GLP-1R; GLP-1-R |
| Ensembl | ENSG00000112164 |
| Gene ID | 2740 |
| mRNA Refseq | NM_002062 |
| Protein Refseq | NP_002053.3 |
| OMIM | 138032 |
| UniProt ID | P43220 |
| Chromosome Location | 6p21.2 |
Clinical Resources
| Gene Function | This gene encodes a 7-transmembrane protein that functions as a receptor for glucagon-like peptide 1 (GLP-1) hormone, which stimulates glucose-induced insulin secretion. This receptor, which functions at the cell surface, becomes internalized in response to GLP-1 and GLP-1 analogs, and it plays an important role in the signaling cascades leading to insulin secretion. It also displays neuroprotective effects in animal models. Polymorphisms in this gene are associated with diabetes. The protein is an important drug target for the treatment of type 2 diabetes and stroke. Alternative splicing of this gene results in multiple transcript variants. |
| Pathway | MAPK signal pathway; cAMP signaling pathway |
| Major Conditions | Diabetes; Cardiovascular disorders; Insulinoma; Myocardial infarction; etc. |
The International Diabetes Federation (IDF) estimated a global prevalence of 463 million adults living with diabetes in 2019, equivalent to 9.3% of the adult (20-79 years) world population, and projected that this number would increase to 700 million (10.9% of adults) by 2045. Diabetes is classified into four major subtypes: type 1, type 2, gestational, and other.
Type 2 diabetes (previously designated non-insulin-dependent diabetes mellitus), or adult-onset diabetes, accounts for over 90% of the diabetic patient population in the Western world. Risk factors for type 2 diabetes include age, obesity, physical inactivity, family history of diabetes, prior history of gestational diabetes, impaired glucose tolerance, impaired fasting glucose, hypertension, polycystic ovary syndrome and race/ethnicity. Type 2 diabetes is strongly favored by genetic predisposition, although environmental factors also contribute significantly to its development.
The objective of diabetes therapy is to achieve the best possible glycemic control while avoiding hypoglycemia, thus reducing the long-term risk of complications. Current treatment guidelines emphasize three major components of treatment of the patient with type 2 diabetes: diet and exercise, normalization of blood glucose levels, and aggressive management of cardiovascular risk factors to prevent micro- and macrovascular complications.
Glucagon-like peptide-1 (GLP-1) is a peptide hormone secreted by intestinal enteroendocrine L-cells, functioning through the combination with GLP-1 receptor. Such combination activates the cyclic adenosine monophosphate (cAMP) pathway and mitogen-activated protein kinase (MAPK) pathway. GLP-1 lowers glucose concentrations by augmenting insulin secretion and suppressing glucagon release. Additional effects of GLP-1 include retardation of gastric emptying, suppression of appetite and, potentially, inhibition of β-cell apoptosis.
The main defects of GLP-1 analogue therapeutics are short duration of action and high incidence of adverse reactions. Ultra-long-acting and with fewer side effects, P11 was developed by applying our unique GLP-1 protection technology to Trulicity (a GLP-1 analogue drug developed by Eli Lilly).
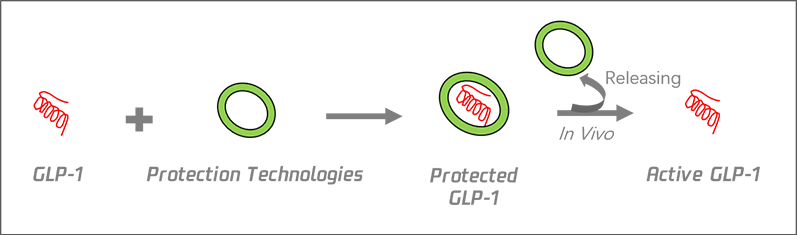
• Prolonging GLP-1 half-life: Preventing degradation mediated by DPP-4 and other proteolytic enzymes.
• Reducing drug side effects/keeping blood concentration constant/expanding application range: Special mechanism to gradually release active GLP-1.
• Further modification of GLP-1: The modified GLP-1 can stimulate the generation of cAMP and stay on the cell surface longer, thus increasing the duration of action to stimulate insulin secretion and reducing the degradation of GLP-1 receptor.
In vitro and in vivo data on P11 are available. Patent application of the protection technology has been submitted.
