| Drug Name | PMB-107 |
| Description |
PMB-107 is a humanized IgG4 antibody that specifically binds to GARP/TGF-β1 complex to block TGF-β1 releasing in the tumor microenvironment. PMB-107 is intended to be used for the treatment of locally advanced or metastatic solid tumors. The candidate is developed in phase I clinical development in the United States. |
| Target | GARP/TGF-β1 complex |
| Drug Modality | Monoclonal antibody |
| Indication | Solid tumors |
| Product Category | Immunotherapy |
| Mechanism of Action | Blocking GARP-mediated TGF-β1 release by binding GARP/TGF-β1 complex |
| Status | Phase I |
| Patent | Granted |
Protheragen Inc. is actively seeking partnership for PMB-107. Potential collaboration can be strategic alliance, licensing, or marketing agreement.
We look forward to hearing from you.
Transforming growth factor-β (TGF-β) subfamily includes TGF-β1, -β2, and -β3, activins A and B, Nodal, and growth and differentiation factors. TGF-β1, -β2, and -β3 are encoded by three different genes and share 71-79% amino acid sequence identity in the C-terminal portion corresponding to mature cytokines. The three isoforms has critical functions in the immune system and especially for regulatory T lymphocytes (Tregs). Among the three types of TGF-βs, TGF-β1 is the most widely expressed in immune cells and is the predominant isoform in the tumor microenvironment.
TGF-βs are synthesized as proproteins that are cleaved in the Golgi apparatus by convertases. The resulting latent form TGF-β (L-TGF-β) is comprised of latency-associated peptide (LAP) and mature TGF-β (mTGF-β), which remain noncovalently associated. Active mTGF-β can be released by interaction between LAP and integrins, including αvβ6 or αvβ8. Alternatively, L-TGF-β can bind to the cell surface molecule glycoprotein-A repetitions predominant (GARP). GARP encoded by LRRC32 is a 72 kDa type I transmembrane protein consisting of 662 amino acids. GARP can activate all three isoforms of L-TGF-β (L-TGF-β1, -β2, and -β3). By binding to L-TGF-β, GARP acts as a docking receptor that concentrates L-TGF-β on the cell surface and enhances their final activation. GARP is expressed on platelets, Tregs, activated B cells, mesenchymal stromal cells, and cancer cells, which suggests that GARP may facilitate an increased local concentration of TGF-β, resulting in TGF-β-dependent immune evasion.
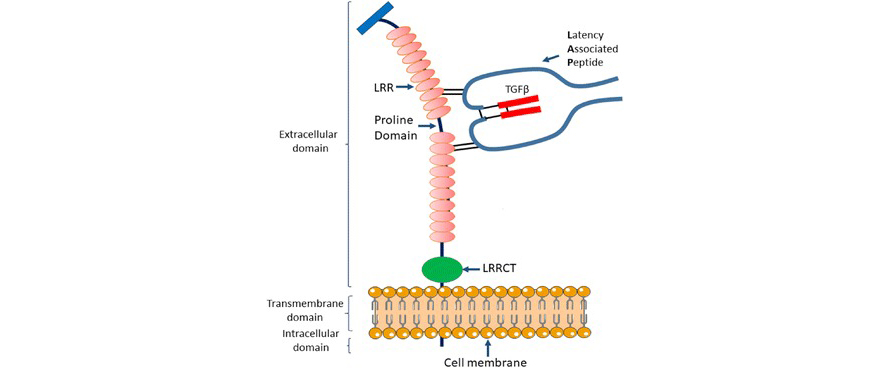 Structure of the membrane-bound GARP/L-TGF-β1 complex
Structure of the membrane-bound GARP/L-TGF-β1 complex
(J Hematol Oncol. 2018;11(1):24.)
PMB-107 is a humanized IgG4 antibody targeting GARP/L-TGF-β1 complex with high affinity, strong specificity, significant anti-tumor efficacy and no toxicity in vivo.
Solid tumors are abnormal masses of tissue that usually do not contain cysts or areas of fluid. Solid tumors may be benign or malignant. Different types of solid tumors are named for the cell type that forms them, such as sarcomas, carcinomas, and lymphomas. There were an estimated 18.1 million cancer cases worldwide in 2020. Of these, 9.3 million cases were in men and 8.8 million in women. Cancer is the leading cause of death worldwide, accounting for nearly 10 million deaths in 2020. The most common cancers and most common causes of cancer death in 2020 were all solid tumors, including breast, lung, colon and rectum, and stomach cancers.
Cancer treatment usually includes surgery, radiotherapy, chemotherapy, hormonal treatments, and targeted biological therapies. Cancer treatment decisions should take into account the stage and biology of the tumor, risk and prognosis of planned therapy, and the economic costs associated with therapy. The global solid tumor cancer treatment market was estimated at $121.3 billion in 2018 and is projected to reach $424.6 billion by 2027, increasing to CAGR by 15.0% from 2019 to 2027.
TGF-β1 exerts multiple activities in tumors. In established tumors, TGF-β1 has pro-tumor functions by promoting epithelial-mesenchymal transition (EMT) and inhibiting the proliferation, differentiation and anti-tumor function of a variety of immune cells. In addition, TGF-β1 contributes to the establishment of immunosuppressive tumor microenvironments by modulating immune and stromal cell functions. Inhibition of TGF-β1 release in the tumor microenvironment is a potential target for precision tumor immunotherapy. The cell surface molecule GARP is the receptor that docks and activates of latent form TGF-β1 and mediates the release of TGF-β1.
By specifically binding to GARP/TGF-β1 complex, PMB-107 blocks the GARP-mediated release of TGF-β1, reverses the immunosuppressive effect in the tumor microenvironment, and improves the anti-tumor immune response. In human tumor cell PBMC humanized tumor animal models, PMB-107 showed tumor growth inhibition. The combination of PMB-107 with anti-PD-1 or anti-PDL1 antibodies showed anti-tumor synergistic effect. In the A375 melanoma xenograft model, PMB-107 combined with anti-PD-L1 antibody induced complete tumor regression in some mice, which was significantly better than that of PD-L1 antibody alone.
Filed PCT application in 2022