| Drug Name | Angiocidin |
| Description |
Angiocidin a 41 kDa recombinant human protein that binds to the TNF superfamily death receptor 6 (DR6) to drive differentiation of leukemic cells and activate the host immune system to fight against acute myeloid leukemia (AML).
As a new therapeutic: |
| Target | DR6 |
| Drug Modality | Recombinant protein |
| Indication | Acute myeloid leukemia |
| Product Category | Immunotherapy |
| Mechanism of Action | Angiocidin, binding to DR6, induces differentiation of leukemic cells and activates host immune system to fight against cancer. |
| Status | Preclinical |
| Patent | US patents have been issued for Angiocidin to treat leukemia and solid tumors. |
Protheragen Inc. is actively seeking partnership to further develop Angiocidin. Potential collaboration can be strategic alliance, licensing, or marketing agreement.
We look forward to hearing from you.
| Introduction |
This gene encodes a member of the tumor necrosis factor receptor superfamily. The encoded protein activates nuclear factor kappa-B and mitogen-activated protein kinase 8 (also called c-Jun N-terminal kinase 1) and induces cell apoptosis. Through its death domain, the encoded receptor interacts with tumor necrosis factor receptor type 1-associated death domain (TRADD) protein, which is known to mediate signal transduction of tumor necrosis factor receptors. Knockout studies in mice suggest that this gene plays a role in T-helper cell activation, and may be involved in inflammation and immune regulation. The deduced 655-amino acid protein has an N-terminal signal sequence followed by 4 TNFR-like cysteine-rich motifs, and a transmembrane domain followed by a cytoplasmic portion containing a 135-amino acid death domain and a 150-residue tail. The death domain of DR6 is 27% identical to that of TNFR1. |
| Approved Name | TNF receptor superfamily member 21 |
| Official Symbol | TNFRSF21 |
| Gene Type | Protein coding |
| Synonyms | TNFRSF21, BM-018, CD358, DR6, tumor necrosis factor receptor superfamily member 21, TNF receptor superfamily member 21 |
| Ensembl | ENSG00000146072 |
| Gene ID | 27242 |
| mRNA Refseq | NM_014452 |
| Protein Refseq | NP_055267 |
| OMIM | 605732 |
| UniProt ID | O75509 |
| Chromosome Location | 6p12.3 |
Clinical Resources
| Gene Function | Within the tumor necrosis factor receptor superfamily, a subgroup, or the death receptor, contains a cytoplasmic death domain. Activation of these receptors leads to the engagement of components of the cell death pathway, including the adaptor molecule TRADD and subsequently the FADD-caspase-8 pathway, which in turn activates the nuclear factor kappa-B pathway. TNFRSF21 belongs to the death receptor subfamily of TNFRs. |
| Pathway | Rho protein geranylgeranylation and nuclear factor kappa B signaling pathway |
Leukemia is heterogeneous class of a malignant diseases of the bone marrow and blood that are characterized by the uncontrolled proliferation of undifferentiated hematopoietic cells. Leukemia is classified as either myelogenous or lymphocytic, according to the type of cell involved. Leukemia is furthermore classified as either chronic or acute, based on the clinical presentation and course.
Acute myeloid (or myelogenous) leukemia (AML) is the most common malignant myeloid disorder in adults, and is associated with the lowest survival rate, although survival has improved steadily in recent decades. It affects more men than women, with a median age of 70 years at diagnosis. In adult patients, the highest rates of AML are seen in North America, Europe, and Oceania, while adult AML is rarer in Asia and Latin America.
The hallmarks of AML are an abnormal proliferation of myeloid progenitor cells in bone marrow, reduced rate of self-destruction and arrest in cellular differentiation. When the blast cells lose their ability to differentiate in a normal fashion and to respond to normal regulators of cell proliferation, the result is frequent infections, bleeding and organ infiltration. The leukemic cells are endowed with an abnormal survival advantage with respect to normal healthy cells, such that the bone marrow and peripheral blood become increasingly populated by immature blast cells that edge out normal blood cells. Bone marrow failure is the most common cause of death in patients with AML.
The main treatment for most types of AML is chemotherapy, sometimes along with a targeted therapy drug. This might be followed by a stem cell transplant. Surgery and radiation therapy are not major treatments for AML, but may be used in special circumstances.
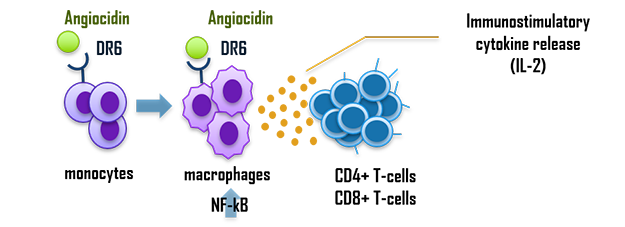
Highly expressed in monocytes and leukemia cell lines, DR6 represents an attractive novel target for AML research. Binding to DR6, Angiocidin drives the NF-kB pathway, induces irreversible differentiation of monocytes into an activated macrophage phenotype, and activated macrophages release cytokines such as IL-2 that activate T-cells. In addition, Angiocidin induces irreversible terminal differentiation in AML cells, reducing the continued progression of the disease and potentiating chemotherapy.
As a new therapeutic for AML, Angiocidin also stimulates AML cell differentiation, reducing the spread of disease and potentiating chemotherapy.
A series of in vivo and in vitro experiments were completed, such as immunoregulatory activity tests and AML xenograft mouse model tests. US patents of Angiocidin treating solid tumors and leukemia have been issued.
