| Drug Name | NP-G2-044 |
| Description |
Fascin is the primary actin-binding protein in filopodia and is critical for actin filament bundling and filopodia formation. High expression of fascin in many metastatic tumors has been suggested as a therapeutic target for blocking tumor cell migration, invasion, and metastasis. NP-G2-044 was identified to specifically inhibit the biochemical function of fascin, which is to bundle actin filaments and to induce the formation of filopodia. Simultaneously targeting tumor cells and dendritic cells within the tumor microenvironment, NP-G2-044 is a novel small molecule to treat different tumors as a single drug or to combine with immune checkpoint inhibitors and chemotherapy. |
| Target | Fascin |
| Drug Modality | Small molecule |
| Indication | Advanced or metastatic solid tumors |
| Product Category | New molecular entity |
| Mechanism of Action | NP-G2-044 occupies the actin-binding site on fascin, thus blocking fascin from binding to actin. |
| Status | Phase 1 completed |
| Patent | Patent applications have been filed in the US, Europe, Canada, and Japan. |
This program is actively seeking investments. We look forward to hearing from you.
| Introduction | Fascin is the primary actin cross-linker in filopodia and has no amino-acid sequence homology with other actin-binding proteins. It functions as a monomer and is required to maximally crosslink the actin filaments into straight, compact, and rigid bundles and to impart distinct mechanical stiffness to actin bundles. Fascin holds 10–30 parallel actin filaments together to form filopodia 60–200 nm in diameter. Three forms of fascin are found in vertebrates: Fascin1, abundant in the nervous system and elsewhere; fascin2 can be found in the retinal photoreceptor cells; fascin3 is only found in the testes. |
| Approved Name | Fascin actin-bundling protein 1 |
| Official Symbol | FSCN1 |
| Gene Type | Protein coding |
| Synonyms | HSN; SNL; p55; FAN1 |
| Ensembl | ENSG00000075618 |
| Gene ID | 6624 |
| mRNA Refseq | NM_003088 |
| Protein Refseq | NP_003079 |
| OMIM | 602689 |
| UniProt ID | Q16658 |
| Chromosome Location | 7p22.1 |
| Gene Function | This gene encodes a member of the fascin family of actin-binding proteins. The expression of this gene is known to be regulated by several microRNAs, and overexpression of this gene has been associated with metastasis of multiple types of cancer by increasing cell motility. |
| Pathway | Fascin bundling actin to form filopodia |
| Major Conditions | Cancer |
NP-G2-044 is a small molecule compound that has been screened and identified from chemical libraries to specifically inhibit the biochemical function of fascin to bundle actin filaments. The optimized fascin inhibitor showed anti-tumor effects in mouse models, blocking the actin-binding and actin-bundling activities of fascin, thus preventing tumor cell migration and metastasis.
|
Fascin expression is low or absent in normal adult epithelial cells. Elevated levels of fascin have been found in many types of metastatic tumors and are correlated with clinically aggressive phenotypes, poor prognosis, and shorter survival. |
| Organ | Subtype | Patients with High Fascin Leve |
| Lung |
Non-small cell lung cancer (NSCLC) stages II+III Squamous cell carcinoma Adenocarcinoma Large cell carcinoma |
63% 98% 78% 83% |
| Breast | Triple-negative | 88% |
| Pancreas | PDAC (pancreatic ductal adenocarcinoma) | 95% |
| Colon | Colonic adenocarcinoma Stages III and IV | 71% |
| Esophagus | Esophageal squamous cell carcinoma (ESCC) | 68% |
| Liver | Poorly differentiated primary hepatocellular carcinoma | 63% |
| Ovary | Stage III/IV ovarian cancer | 53% |
| Lymphoma | Hodgkin lymphoma | 100% |
|
Lung cancer is the deadliest cancer in the world. In 2018, the total number of deaths from lung cancer is 1.76 million, equivalent to 18.4% of all cancer deaths. Breast cancer is the most commonly occurring cancer in women and the second most common cancer overall. About 10-20% of breast cancers are triple-negative breast cancers that does not respond to hormonal therapy medicines or medicines that target HER2 protein receptors. Pancreatic cancer is the fifth leading cause of cancer deaths following breast cancer, lung cancer, colon cancer, and prostate cancer. Pancreatic ductal adenocarcinoma (PDAC), which constitutes 90% of pancreatic cancers, is one of the most aggressive and lethal cancers with very few available treatments. The median survival of PDAC patients was about 6 months after diagnosis. By 2030, PDAC is projected to become the second leading cause of cancer deaths in America, second only to lung cancer. |
NP-G2-044 could occupy the actin-binding site on fascin to change the conformation of fascin, reducing the binding of actin filaments. Fascin is the main actin-bundling protein in filopodia, which are fingerlike plasma membrane protrusions that are formed upon remodeling of the actin cytoskeleton beneath the plasma membrane. Viewed as a sensory organ of the cells, filopodia are used to detect and assimilate signals as well as to explore and move into the surrounding microenvironment. Metastatic tumor cells are rich in filopodia, and the numbers of filopodia correlate with their invasiveness.
Through monitoring the signaling systems downstream of fascin, NP-G2-044 was found to block filopodial formation, tumor cell migration, invasion, and metastasis. In addition, NP-G2-044 also activates intertumoral dendritic cells and increases antigen uptake, so it could be used in combination with immune checkpoint inhibitors.
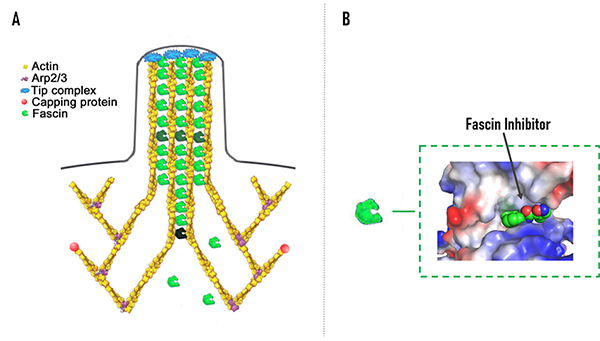
Figure A. from J Cell Biol (2006) 174 (6): 863–875.
Patent applications have been filed in the US, Europe, Canada, and Japan.

Clinical data can be viewed after a signing a CDA. Initial efficacy was observed in multi-centered phase I trials.