| Drug Name | Anti-CD40-7409 |
| Description |
Anti-CD40-7409 is a humanized agonistic monoclonal antibody targeting CD40 with an engineered Fc capable of selectively binding to FcγRIIB. Anti-CD40-7409 activates CD40 in Fc-FcγRIIB cross-link dependent manner and therefore operates optimally in a CD40 and FcγRIIB-enriched tumor microenvironment. Anti-CD40-7409 is in early clinical development for the treatment of advanced or metastatic solid tumors, both as monotherapy and in combination with other therapies. |
| Target | CD40 |
| Drug Modality | Monoclonal antibody |
| Indication | Advanced or metastatic solid tumors |
| Product Category | Cancer immunotherapy |
| Mechanism of Action | FcγRIIB mediated CD40 activation |
| Status | Phase 1 |
| Patent | Granted |
Protheragen Inc. is actively seeking partnership for Anti-CD40-7409. Potential collaboration can be strategic alliance, licensing, or marketing agreement.
We look forward to hearing from you.
| Introduction | CD40 is a 48 kDa type 1 transmembrane protein that expressed on antigen-presenting cells (APCs) including dendritic cells (DCs), B cells, macrophages, and classical and non-classical monocytes, and on a variety of non-immune cells including platelets and endothelial cells. The binding of CD40 and CD40 ligand (CD40L) plays a crucial role in both adaptive and innate immunity and engages signaling pathways that are involved in maturation, proliferation, and survival of DCs, production of inflammatory cytokines, and expression of costimulatory molecules. |
| Approved Name | CD40 molecule |
| Official Symbol | CD40 |
| Gene Type | Protein coding |
| Synonyms | p50; Bp50; CDW40; TNFRSF5 |
| Ensembl | ENSG00000101017 |
| Gene ID | 958 |
| mRNA Refseq | NM_001250 |
| Protein Refseq | NP_001241 |
| OMIM | 109535 |
| UniProt ID | P25942 |
| Chromosome Location | 20q13.12 |
| Gene Function | This gene is a member of the TNF-receptor superfamily. The encoded protein is a receptor on antigen-presenting cells of the immune system and is essential for mediating a broad variety of immune and inflammatory responses including T cell-dependent immunoglobulin class switching, memory B cell development, and germinal center formation. |
| Major Conditions | Cancers; Autoimmune disease; Transplant rejection |
Based on a proprietary monoclonal antibody functional platform, Anti-CD40-7409 is developed with tumor-localized immunostimulatory activities by balancing multiple functions of antibodies. Anti-CD40-7409 is an agonistic IgG antibody that activates CD40 optimally in the presence of FcγRIIB to increase selectivity for tumors. FcγRIIB is expressed on immune cells enriched in the tumor microenvironment, including B cells, macrophages, dendritic cells, monocytes, and NK cells. The higher expression profile and level of FcγRIIB in tumor tissues relative to normal tissues supports robust antitumor efficacy and superior safety profile of Anti-CD40-7409 driven by Fc-FcγRIIB cross-linking.
Solid tumors are abnormal mass of tissue that usually does not contain cysts or liquid areas. Solid tumors may be benign or malignant. Different types of solid tumors are named for the type of cells that form them, such as lung cancer and breast cancer. According to the WHO Global Cancer Observatory (GLOBOCAN), lung cancer (1.80 million deaths), colon and rectum cancer (916,000 deaths), and liver cancer (830,000 deaths) were the three most deadly cancers in 2020. 18.09 million new cases of cancer had been diagnosed in 2020 in which breast (2.26 million cases), lung (2.21 million cases), and colon and rectum (1.93 million cases) being the three most frequent.
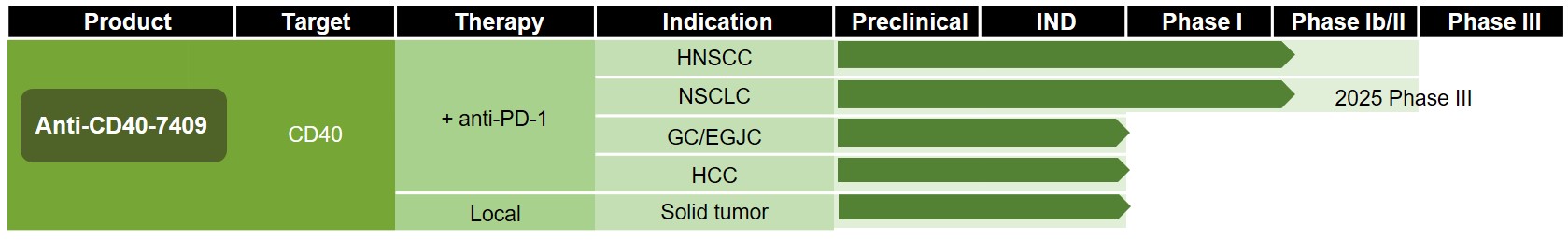
Anti-CD40-7409 monotherapy and in combination with anti-PD-1 antibody therapy are in early clinical development for the treatment of advanced or metastatic solid tumors.
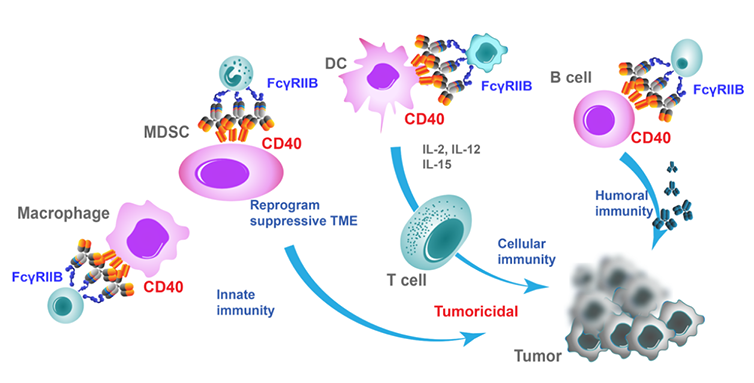
CD40 is a costimulatory protein expressed on antigen-presenting cells (APCs) and its interaction with the CD40L plays an important role in the amplification of the immune response and the production of antibodies. Similar to the endogenous CD40L, agonistic antibody Anti-CD40-7409 binds to CD40 to activate APCs and induce a variety of downstream effects, such as the cellular proliferation, activation, and antigen presentation of B cells, and activation of antigen-specific T cells. This results in an enhanced immune response against tumor cells.
The modified Fc fragment of Anti-CD40-7409 retains residual binding to FcγRIIB. Anti-CD40-7409 activates CD40 signaling in an Fc-FcγRIIB cross-link dependent manner and therefore operates optimally in a CD40 and FcγRIIB-enriched tumor microenvironment.
Phase I trials of Anti-CD40-7409 monotherapy and combination therapy with anti-PD-1 antibody are nearing completion.