| Drug Name | OV-FV-01 |
| Description |
OV-FV-01 is a genetically modified oncolytic flavivirus. It infects and kills cancer cells, and also activates T cells by promoting cancer cells to produce double signals of tumor antigen and costimulatory factors, thus killing cancer cells. Because flavivirus is susceptible to a variety of epithelial cells, OV-FV-01 has a broad spectrum of anticancer properties and can be used on cancers that cannot be treated by conventional therapies, such as lung cancer, stomach cancer, head and neck cancer, and melanoma. Significant inhibition of tumor growth was observed in groups of mice with lung and gastric cancers treated with OV-FV-01. Total effective rate was determined to be 65%. |
| Target | Confidential |
| Drug Modality | Oncolytic virus |
| Indication | Solid tumors, such as lung cancer, stomach cancer, head and neck cancer, and melanoma. |
| Product Category | Immunotherapy |
| Mechanism of Action | OV-FV-01 directly lyses tumor cells, leading to the release of soluble antigens, which drive antitumor immunity. In addition, OV-FV-01 is engineered to express costimulatory molecule, further enhancing T cell activity. |
| Status | Preclinical |
| Patent | Related patents have been obtained. |
Protheragen Inc. is actively seeking partnership to further develop OV-FV-01. Potential collaboration can be strategic alliance, licensing, or marketing agreement.
We look forward to hearing from you.
Because flavivirus is susceptible to a variety of epithelial cells, OV-FV-01 has a broad-spectrum of solid cancer indication, including lung cancer, gastric cancer, melanoma, etc.
| Lung cancer |
Lung cancer is the most prevalent and costly malignancy in the world today, as well as the most frequent cause of cancer-related mortality in both men and women worldwide. The two most common forms of lung cancer are non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). Patients presenting with symptoms indicative of lung cancer should undergo a thorough diagnostic workup including evaluation of the patient’s medical history, smoking history, exposure to environmental and occupational carcinogens and family history of cancer. A complete physical examination should be performed. Computer tomography (CT) scan of the chest is indicated in all patients. Unfortunately, many patients report a significant delay between emergence of symptoms and diagnosis of lung cancer. According to the International Agency for Research on Cancer (IARC), lung cancer is the malignancy of highest impact in the world today, both in terms of the total number of individuals it affects (2.1 million new cases in 2018, representing 11.6% of all new cancers) and the total number of resulting deaths (1.76 million in 2018, equivalent to 18.4% of all cancer deaths). |
| Gastric cancer |
Gastric cancer comprises a number of malignancies arising in the tissues lining the stomach, anywhere between the gastroesophageal junction and the pylorus. Adenocarcinomas, or cancers arising in epithelial cells, account for approximately 95% of all gastric cancers that can metastasize to the liver, peritoneum and lymph nodes. The diagnostic workup of a patient suspected of having stomach cancer includes obtaining a complete medical history and physical examination, as well as performing an upper GI endoscopy for visualization of the gastrointestinal tract and for obtaining biopsies. According to the IARC, there were more than one million new cases of stomach cancer diagnosed in 2018, making it the fifth most common malignancy in the world. Based on incidence data from the Globocan 2008 database and projected population increases, the number of new cases is predicted to exceed 1.7 million in 2030. There were 782,685 deaths from stomach cancer worldwide in 2018. Stomach cancer was the third leading cause of cancer-related death worldwide in the same year. |
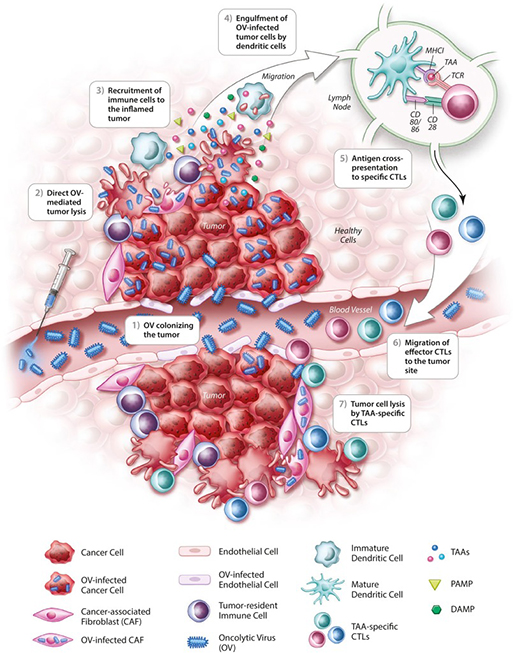
The emerging oncolytic virus (OV) therapy is a promising treatment strategy for malignant tumors. OVs can achieve the goal of targeted therapy by directly killing cancer cells and inducing antitumor immune response. OV treatment refers to the use of a virus to self-replicate and destroy cancer cells by infecting and lysing those cells. The infected cancer cells continue to release virions, or new virus particles, annihilating the rest of the cancer cells. To escape the immune surveillance, cancer cells secrete certain factors to inhibit the immune system. But OV has a strong stimulating effect on immune cells infiltrating the tumor tissue, which can greatly alter the tumor microenvironment. Infected tumor cells are able to express cytokines, which can induce immune cells outside of the tumor to infiltrate and activate non-specific immune cells. Tumor cells lysed by OV release large amount of tumor proteins that can be phagocytized by non-specific immune cells, and certain tumor-specific antigens can be expressed by these antigens presenting cells, inducing T cells to attack uninfected tumor cells.
According to the process during development, OVs can be divided into natural viruses and genetically modified virus strains. OV-FV-01 is a genetically modified oncolytic virus belong to flavivirus, a genus of viruses in the family Flaviviridae. The flavivirus family shares several common aspects: 40–65 nm in diameter, symmetry of enveloped, icosahedral nucleocapsid, single-stranded RNA around 10,000-11,000 bases. In addition to its natural ability to infect cancer cells, OV-FV-01 is also genetically modified to express co-stimulating molecules that enhance the immunity of T cells.
EBioMedicine 31 (2018) 17–24
● Related patents have been obtained.
● Virus construction, small-scale production, quality testing for 6 plasmids of mouse immune factors and effective tests in vivo have been completed.
